Regional
Tuberculosis kills more people than malaria or HIV. Why haven’t we found a vaccine?
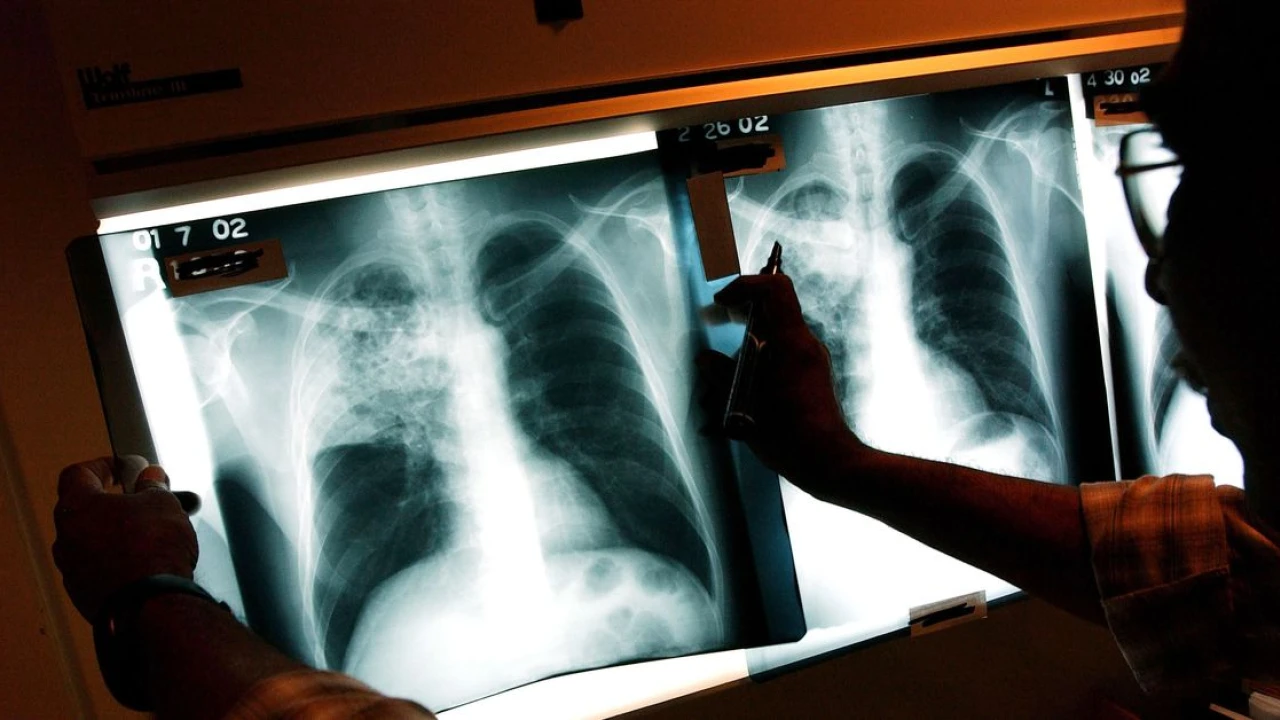
More than a million people die annually from tuberculosis, a bacterial lung disease. Volunteers could speed up the race for a TB cure that works for adults as well as children.
Few forces have killed off talented people before their time with quite the effectiveness of tuberculosis. There’s Chekhov, Chopin, Orwell, Kafka, Keats, Thoreau, Emily Brontë — all brought down by the bacterium before they reached the age of 50.
Their ranks are still growing. About 1.3 million people died of TB in 2022, and while deaths were falling pre-pandemic, the Covid-19 era saw progress stall. At this point, its annual death toll exceeds that of HIV/AIDS or malaria. The one vaccine against it was created in 1921 and does not protect adults or adolescents.
Yet the world has not mustered much in the way of resources against the disease. The New York Times’s Stephanie Nolen notes that $5.8 billion a year in treatment funds and $1 billion a year in research funds goes to fighting TB in low- and middle-income countries. By contrast, the equivalent figure for HIV/AIDS treatment is about $20 billion annually. That’s a worthy cause, of course, but considering its enormous death toll, TB has drawn the short straw.
Why is TB so neglected?
To some degree, the relative neglect of TB (including by me — I write much more about malaria) has to do with the scale of the challenge it poses.
TB is a hard disease to vaccinate against. While most vaccines target viruses, TB is a bacterium, and one with a strange lifecycle. In the vast majority of people it infects, it is “latent,” not causing symptoms or becoming contagious. The population of people carrying around latent TB infections is truly massive. A recent study compiling blood test estimates found that about 24 percent of people on Earth have TB, with rates ranging from 12–14 percent in Europe and the Americas to over a third in Africa and Southeast Asia.
A small share of people with these latent infections (from 5 to 15 percent) develop symptomatic, contagious cases. Often these are treatable with antibiotics — but a significant share of cases are drug-resistant, access to antibiotics is uneven in low-income countries, and mortality is particularly high in people with both HIV and TB.
These dynamics complicate the task for a vaccine. Ideally you would want it to both reduce the share of people with latent infections and reduce the odds that those infections become active. But we don’t have what’s called a “correlate of protection” for TB: a set of indicators of a person’s immune system that show they can resist initial infection, or can keep an infection from becoming active.
That makes testing treatments and vaccines tricky — and expensive. Testing needs to be truly massive in scale, given the relatively small share of people who gain new latent infections, or see infections go active, in a given year. M72, the most promising vaccine candidate as of right now, is currently undergoing a phase III trial with a staggering 26,000 participants. It cost donors $550 million to fund it — more than half the annual research budget for TB. And there’s still a chance it won’t work, or even that the trial won’t pick up enough cases to show anything either way.
If every promising TB vaccine costs more than half a billion dollars to test, we are not going to test very many. Even if M72 works, and I very much hope it does, it may still have limitations, as the existing vaccine for children does; and there may be other models that are cheaper to manufacture, or that prove more effective. We want to be testing those, too, and we won’t if testing them is prohibitively expensive.
How to make vaccine testing cheap
In 2016, the FDA approved a cholera vaccine whose phase III test only included 197 people. That’s a tiny fraction of the 26,000 in the TB vaccine trial. This earlier trial was much, much cheaper but still resulted in a working, approved vaccine.
What was the trick? The cholera vaccine was assessed using a “challenge” trial. 134 of the volunteers in the study actually ingested the cholera bacterium after receiving their vaccine or placebo. Those who got infected were, of course, given antibiotics, fluids, and other necessary treatments. No one got hurt. But their sacrifice enabled researchers to find an effective cholera vaccine for a fraction of the price of a field trial with thousands of people.
Human challenge trials are a tested and reliable method, having been used for decades with great success to develop malaria treatments and prophylactics. But they have yet to be used on tuberculosis. Developing a challenge approach takes time, and while that investment has been made for malaria and cholera, it hasn’t been for TB yet. That’s a shame, because they could provide a way around the huge numbers of people TB trials typically need to detect results. Instead of waiting months for a handful out of a group of thousands of participants to be exposed to TB, researchers could expose several dozen volunteers at once, see how much protection vaccines offer, and proceed from there.
This may sound dangerous at first glance, but the risks are small. As with other challenge trials, the researchers would use a weakened or “attenuated” version of the pathogen and would provide immediate and comprehensive treatment. One paper examining the idea estimated that a challenge trial would pose a risk of death of about 1 in 1,600, at the high end. For context, that’s less than half the death risk associated with working as a trucker for five years, and about one-sixth the danger of working as a logger for five years. We let people do those things for money because we value lumber and quick shipping. There’s something to be said for letting people take more modest risks to save lives.
Luckily, several researchers are trying to develop models for tuberculosis challenges. One would use the 1921 vaccine itself as a substitute for the bacterium, because the vaccine is already in essence an attenuated form of TB. Given that the vaccine is already broadly accepted, it seems hard to argue with the acceptability of this approach. Another option involves an artificial strain of tuberculosis that has been modified to rely on certain compounds that can be withdrawn at any time, effectively offering a “kill switch” for the bacteria and allowing study researchers to fully cure participants at the end of the trial. Both of these could provide a pathway to challenge trials with extremely minimal risks for participants.
Helen McShane, Eric Rubin, Sarah Fortune, and the dozens of other researchers developing these models are, I think, doing some of the highest-impact work in global health right now. But they need help: regular funding, collaborative drug agencies, and a general public that’s supportive of letting volunteers contribute to the fight against tuberculosis this way.
A version of this newsletter originally appeared in the Future Perfect newsletter. Sign up here!
-

 Regional 1 day ago
Regional 1 day agoPPP wins 8 seats in Karachi by-elections
-

 Pakistan 10 hours ago
Pakistan 10 hours agoCourt reserves verdict in Sharjeel Memon's assets case
-

 Pakistan 1 day ago
Pakistan 1 day agoLucky numbers of Rs100, Rs1,500 prize bonds announced
-

 Sports 2 days ago
Sports 2 days agoIndian Kabaddi team not allowed to visit Pakistan
-

 Business 1 day ago
Business 1 day agoGold price shoots up Rs1,300 per tola in Pakistan
-
 Technology 1 day ago
Technology 1 day agoNew ‘Draft’ feature in WhatsApp
-

 Business 1 day ago
Business 1 day agoGeneral Motors lays off about 1,000 workers to compete in a crowded automobile market
-

 Crime 2 days ago
Crime 2 days agoTwo kids killed as device explodes in N. Waziristan